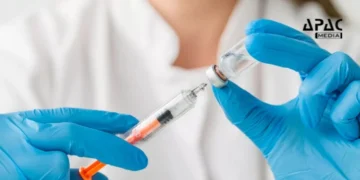
New Delhi: Malaria claims approximately half a million lives every year, globally. However, this number has a possibility of seeing a steady decline after WHO’s approval to globally roll out R21/Matrix-M, the malaria vaccine developed by the University of Oxford and the Serum Institute of India.
The R21/Matrix-M has now been recommended by the World Health Organisation’s Strategic Advisory Group of Experts and the Malaria Policy Advisory Group. It leverages Novavax’s adjuvant, and is to be supplied to countries burdened with malaria outbreaks, the University of Oxford said.
The Serum Institute of India has currently set up a production capacity of 100 million doses per year, and they plan to increase this capacity twofold within the next two years, according to the Oxford University website. Adar Poonawalla, chief executive officer of the Serum Institute of India, said Serum’s aim is to increase vaccine production to make it accessible to the people who need it the most.
“For far too long, malaria has threatened the lives of billions of people across the globe, disproportionately affecting the most vulnerable amongst us,” Poonawalla said. He said the approval of R21/Matrix-M represents a significant milestone on the journey to combat this life-threatening disease, demonstrating what can be achieved when the public and private sectors, scientists, and researchers all collaborate towards a common goal.
Also Read More –
- Cash-Strapped Edtech Giant BYJU’S Announces Major Workforce Restructuring, 4,000 employees to be laid off
- IIT Council inks MoU with Association of American University for Indo-US Global Challenges Institute



















































Discussion about this post